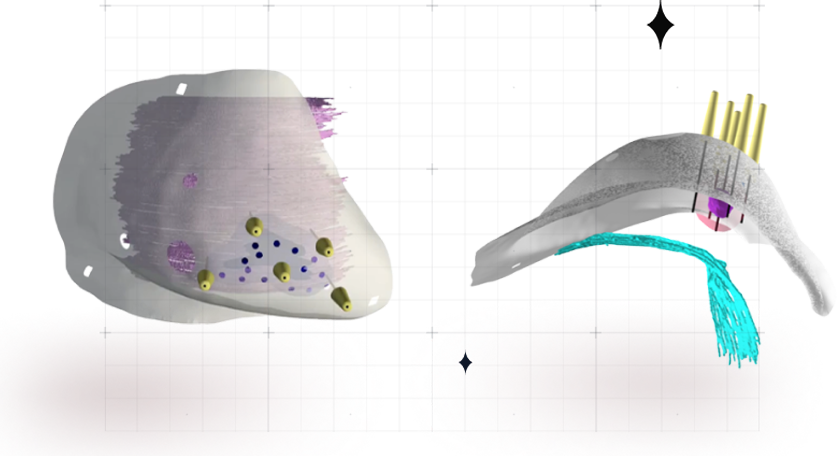
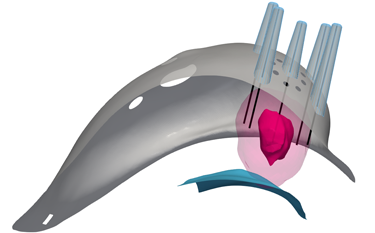
The Visualizer™
The Visualizer™ provides an interactive 3D view of the area surrounding the tumor of the patient in the supine position –
the same position the patient is in on the Operating Room table.
For the first time, surgeons are using 3D visualization to localize and direct total breast cancer tumor removal.